



LPS: the most potent single inducer of inflammation
Over centuries, clinical observations indicated the presence of a substance in water, injection fluids and body fluids that could cause fever and even death.Understanding lipopolysaccharides
The mysterious substance, initially considered a fever-inducing compound, was associated with Gram-negative bacteria. Investigations showed that it was a major component of the bacterial cell wall, eventually described as endotoxin, to clearly discriminate it from other bacterial toxins, secreted by bacteria into their environment.
It was initially believed that an endotoxin could only be liberated form the cell wall when Gram-negative bacteria die. Current insights reveal, however, that endotoxins are also shed by living Gram- bacteria during normal physiological processes such as cell replication. The actual composition of the polysaccharides varies between bacterial species, and the substance is now generally described as lipopolysaccharide (LPS).
The origin of LPS explains many of the clinical symptoms in the intestines and the liver of all animals and humans, but also in specific organs.
The LPS challenge
Animals are exposed to the LPS via different routes.
Feed and water will always contain some amounts of LPS, which might reach critical levels when the microbiological quality of the feed is very poor. Processing may reduce the bacterial load but fails to inactivate the heat-stable LPS.
LPS also adheres to fine dust particles in the air of animal houses and can result in significant respiratory exposure.
The major natural source of LPS, however, is the complex community of Gram-negative bacteria in the intestines.
An intact intestinal barrier can protect against LPS
Several natural defence mechanisms, as part of the innate immunity, are in place to protect the body. For example, intestinal cells express the enzyme alkaline phosphatase that can inactivate LPS.
Moreover, in an intact intestinal barrier, the epithelial lining cells are crosslinked by a network of tight junction proteins, making the cell layer impermeable for larger molecules like LPS.
Stress factors impair intestinal defences
The intestinal defence mechanisms, however, can be impaired by various internal and external factors. These include acute viral, bacterial, or protozoal infections as well as dietary exposure to toxins like mycotoxins.
In addition, stress conditions, like heat stress or even feed or water deprivation can lead to a loss of function of the intestinal barrier. In these cases, intestinal LPS will reach the systemic circulation and initiate a cascade of adverse effect.
The cascade of adverse effect in animals induced by LPS
LPS is recognised by different classes of immune cells as a danger molecule. LPS recognition by neutrophils, macrophages or dendritic cells in the intestines, the liver and other tissues involves TLR4 receptors. Activation of these receptors initiates a complex cellular response, resulting in the production of pro-inflammatory mediators such as cytokines, prostaglandins, nitric oxide, and other oxygen radicals (ROS).
The cytokine IL-6 dysregulates body temperature control, measurable as febrile response. Depending on the amount of LPS, the elevation of body temperature is gradually visible as a mild fever or as extreme high body temperature, resulting in a fatal shock syndrome.
At tissue level, the inflammatory mediators induce the typical symptoms of inflammation: redness, oedema, pain and dysfunction.
In the intestines, an inflammatory response is often characterized by diarrhoea, with excessive loss of water and electrolytes, reduced nutrient utilization, and the loss of barrier function, thereby maintaining the fatal LPS challenge.
Mitigating the LPS challenge
Numerous compounds have been tested in the past for their ability to mitigate the adverse effects of LPS. These include certain anti-inflammatory drugs and LPS-inactivating agents, like recombinant alkaline phosphatase.
Unexpected help
Recently, specific products that have been tested for their ability to bind mycotoxins have shown to also bind LPS. To prove this effect, assays were conducted in in vitro systems, in which LPS activity reduction is measured by the LAL assay, comparing several products.
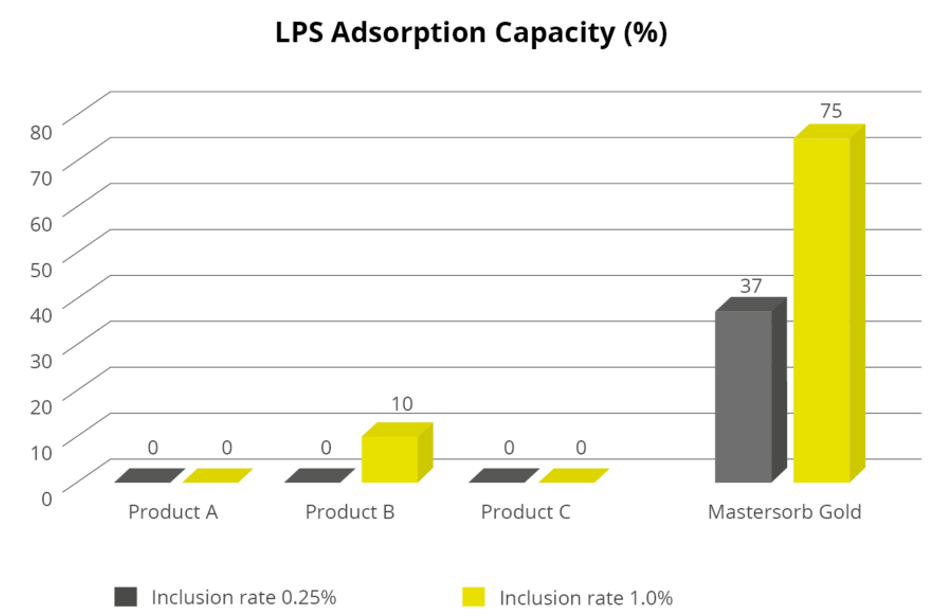
These promising in vitro results demonstrate the efficacy of specific products and indicate the opportunity of using such products as a practical and economically viable measure to reduce the LPS challenge under conditions of stress and/or mycotoxin exposure in animals.








