



The next generation of water-soluble probiotics, providing a possible alternative to AGPs
Antibiotics have been used for decades as growth promoters (AGPs), and they continue to be employed in many countries despite being suspected of supporting the development of antimicrobial resistance.In its Global Action Plan on Antimicrobial Resistance, the World Health Organization (WHO) has called for prudence in administering drugs to animals and expressed concerns over their increased use in animal production, even for therapeutic purposes. Many feed additives have been developed as alternatives to AGPs due to a focus on maintaining the gut homeostasis. Probiotics have been shown to be an efficient feed additive to maintain intestinal integrity and strengthen the immunity of the bird, thus reducing the need for repeated medication. As feed additives, probiotics are included in the feed and administered continuously during the entire production cycle.
Water-soluble gut health stabilizers are often applied to quickly react to upcoming stress situations in the flock – but they can negatively impact the formation of biofilm in the water lines. This is one of the reasons why an alternative administration tool for probiotics through the water supply has been developed. The new formulation has the advantage of quick and easy handling characteristics and can be supplemented at short notice to try and support animals in case of unexpected challenges. In this article we will describe the specifics of this new probiotic formulation and its advantages in vitro and in vivo.
1. New formulation and its advantages
The innovative product is an effervescent tablet loaded with a natural, fast-growing Bacillus amyloliquefaciens CECT 5940. The production is carried out in two main steps. The individual raw materials of the formulation are mixed and subsequently transferred to the tablet press, where the tablets are created and automatically packed into individual sachets by the packaging unit.
Each round speckled tablet has a rough surface and a beige to light brownish color. It weighs around 13 grams and has a diameter of 41 mm (see Figure 1).

In addition to an excellent dissolution rate, the effervescent effect ensures a homogeneous distribution and release of the probiotic spores in water without an additional stirring step. After dissolving the tablet, which requires an average of 10 minutes, the spores are immediately distributed homogeneously in water and remain there for several hours. In general, the tablet offers an extremely customer-friendly product form. It makes handling probiotics easier by simplifying the dosage and reducing the time needed compared to similar products on the market. With only one tablet in 1,000 liters, the desired spore concentration of 5E+8 colony-forming units (CFU) per liter can be achieved easily and repeatedly. Of course, a more concentrated stock solution can also be prepared without any problems by using a lower starting volume.
This water application guarantees flexibility, especially in stressful periods, regardless of the current feed formulation. Moreover, the concentrated probiotic effervescent tablet is compatible with other feed additives – like vitamins, phytogenics and various organic acids – in several concentrations. The tablet is also still effective during the application of disinfectants, such as hydrogen peroxide. Different water qualities do not impact the sparkling activity of the tablet or the desired efficacy of the ingredients, particularly the active probiotic strain.
Based on the unique sugar-free composition of the formulation, the product’s ability to prevent biofilm formation was a particular highlight of its application in water supply lines. Ecobiol® Fizz forms less biofilm than other commercial water-soluble probiotics with dextrose. This finding is supported by data from a biofilm assay carried out in the laboratory. These tests were carried out in close accordance with conditions as applied in farms. In addition to the choice of material (PVC and stainless steel), the flow rates of the water were adjusted depending on the growth period and the changed drinking behavior of the animals. Over a total period of 42 days, fresh water with the additional probiotic products was prepared daily and circulated at a target concentration of 5E+8 CFU per liter.
After 42 days, the hoses were removed from the test system and dyed with crystal violet for a defined exposure time to detect the organic residues of biofilm. For photometric measurements, the stained hoses were rinsed with an ethanolic solution and violet-colored solutions were collected. The photometric determination allows conclusions to be drawn on the intensity of the formed biofilm in the water pipes. PVC is much more sensitive to biofilm formation than stainless steel. The results show that the innovative effervescent tablet reduced biofilm formation in steel tubes by up to 38 percent and in PVC tubes by up to 46 percent compared to commercial probiotics with dextrose.
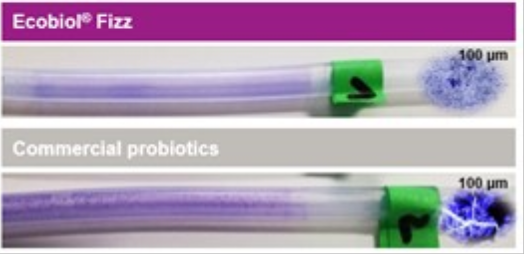
Furthermore, microscope images show clear differences in the size of the particles formed in the hose (see Figure 2). Crystal-violet stained biofilm-particles are much bigger in the drinking pipes containing commercial probiotics with dextrose compared to the new effervescent tablet without a sugar-based carrier or additives.
2. Animal feeding trial
The probiotic formulation in form of an effervescent tablet (Ecobiol® Fizz) was tested under field conditions in a commercial farm. The farm, located in the UK, was composed of six houses of 36,500 birds each. The different houses were randomly allocated to the different treatments: three control houses (C) and three houses received the tablets in water (E). One tablet was dissolved in a 20 L stock solution and then supplemented to the drinking line at a concentration of 2%. The target is to have one tablet/1,000L and a final concentration of 10E+8 CFU/L of water. The tablets were administered on an intermittent model during a stress period. The birds received the probiotic in drinking water for three days after hatch and during the feed transition for approximately 12 hours/day for three days to follow. The thinning was applied at day 32, when only 20,000 broilers were left in the house and the rest sent to the slaughterhouse.
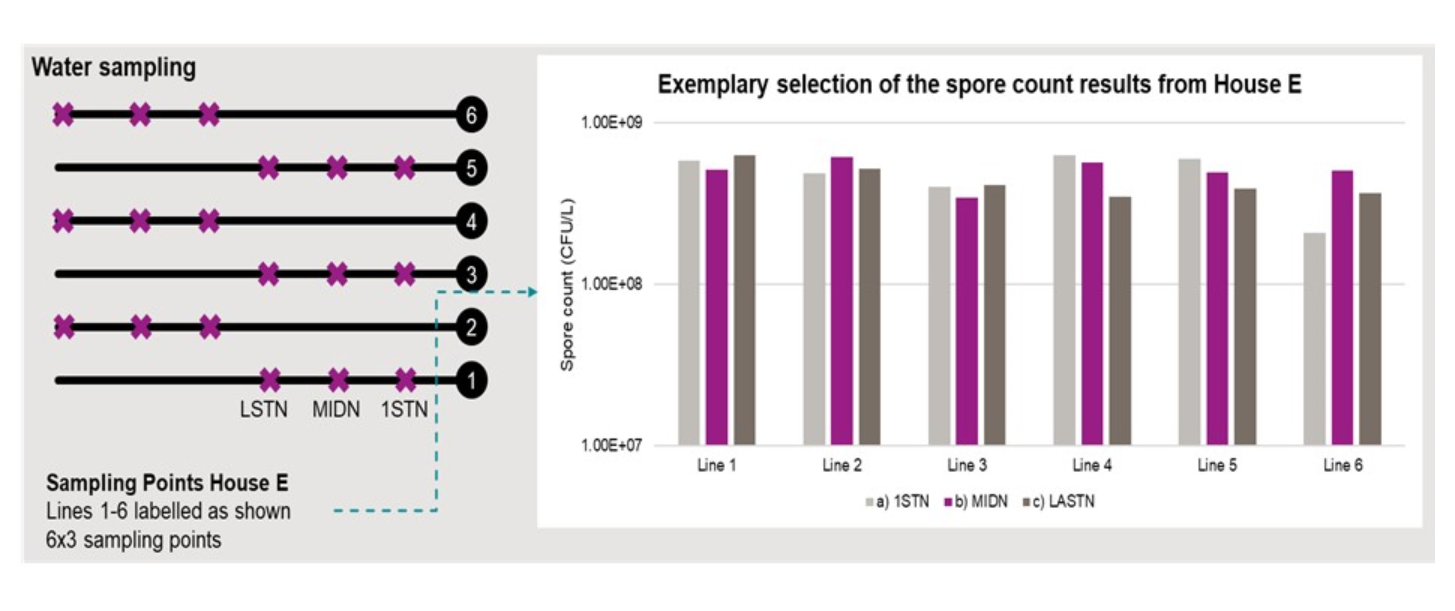
Water samples were taken at different positions in all drinking lines 1-6 and during all application periods – first drink nipple (1STN), middle drink nipple (MIDN), and last drink nipple (LASTN) (see Figure 3).
Spore count was performed on these samples and results showed a homogenous distribution of the probiotic along and between drinking lines (Figure 4) but also during the different supplementation periods, even during the starter period when water consumption was lowest.
Animal performance: feed and water consumption, average body weight (Av BWG), feed conversion ratio (FCR) and mortality were recorded daily during the entire rearing period. Average results for the control and treatment houses, including slaughterhouse parameters (hock burn, pododermatitis and factory rejections) are presented in Table 1.
The performance of the treatment group and the control houses are detailed in Table 1.
Parameters |
Control |
Ecobiol® Fizz |
|
|
EPEF |
414.49 |
414.04 |
|
|
Av BWG (kg) |
2.230 |
2.264 |
|
+34 g/bird |
FCR |
1.448 |
1.495 |
|
+0.04 |
Mortality % |
6.51 |
5.31 |
|
-1.2 % |
Hock % |
29.72 |
24.58 |
|
-5.14 % |
Podo % |
25.30 |
19.82 |
|
-5.48 % |
Factory rejects % |
0.49 |
0.38 |
|
-0.1% ~1095 Birds |
Total weight (kg) |
230 103 |
235 157 |
|
+ 5 054 kg |
Total weight € |
345 155 |
352 736 |
|
+ 7 581 € |
Total feed intake (kg) |
333 350 |
352 270 |
|
+18 920 kg |
Total feed intake € |
76 671 |
81 022 |
|
+ 4 352€ |
ROI |
5.44 |
|||
Applying the new formulation of the probiotic has shown increased body weight in the treatment houses compared to the control houses. Hock burn and pododermatitis scores were improved by approximately 5% in all three treatment houses as well as less factory rejects compared to the control. The lower incidence of hock and pododermatitis in the treatment houses may be attributed to the lower moisture content of the litter as indicated by the decrease in feed: water ratio (1.85 and 1.78 for C and E, respectively). During the trial, two houses (C and E) suffered badly from chronic yolk sac infection. On day three, a vet attended the site and reported 3.39% mortality in house C and made the decision to medicate with antibiotics. The vet also stated the probiotic had a protective effect on the birds and mitigated the losses on house E (reported only 0.82% mortality at the same point), avoiding the necessity for antibiotic treatment. This is significant as these two houses were from the same flock and the same parent flock, and assuming that they would have been equally affected by the hatchery related issues leading to the subsequent development of retained yolk sac.
In general, the tablet was well received by the end user who benefited from easy and quick handling. It also showed a positive return on investment. Additionally, an independent vet opinion affirmed the probiotic had a beneficial protective effect on the chick gut health during infection. This may prove to be an alternative to antibiotic use and produce a subsequent reduction in antimicrobial resistance. The probiotic authenticates a fast and convenient solution for farm supplementation of probiotics in water, without a big impact on biofilm formation in the drinking lines.
Conclusion
Efficient poultry production is a balancing act between various factors, with nutrition and gut health playing a crucial role. The probiotic effervescent tablet, Ecobiol® Fizz, can be applied conveniently on-demand via water supply lines to rapidly improve the composition and balance of the intestinal microbiota. Feeding trials demonstrated that livestock performance and ROI increased with the use of the tablet. The easy handling and the broad product compatibility with other feed additives in various water conditions emphasizes the customer-friendly aspects and the product benefits. As well as these advantages, the formation of a biofilm is significantly reduced with this new formulation.









