



Improving Cardiopulmonary Performance in Broilers Chickens through Nutrition
The concurrent supplementation of arginine, vitamin E and vitamin C improved the pulmonary vasodilation of broiler chickens grown under cold environments after an acute epinephrine challenge, according to Dr Ciro A. Ruiz-Feria of Texas A&M University, USA.Introduction
Pulmonary hypertension syndrome (PHS, ascites) is a common metabolic disorder of fast growing modern strains of broilers. Ascites mortality peaks when the birds are five to six weeks old, which represents a significant loss to the industry given the cost of production up to this point (Maxwell and Robertson, 1998). Ascites can be initiated by an increase in metabolic rate (rapid growth or cold stress) which implies higher oxygen demands (Buys et al., 1999; Scheele et al., 1991; Wideman and Tackett, 2000) and higher amounts of blood must be propelled through the non-compliant pulmonary vasculature, increasing pulmonary arterial pressure (PAP) and right ventricle work (Wideman, 1988). Sustained or chronic increases in PAP cause right ventricular hypertrophy and dilation of the right ventricle (Peacock et al., 1990; Wideman, 1999) leading to right ventricular failure and death.
By reducing the pulmonary vascular resistance, it is possible to reduce the PAP needed to propel the cardiac output required to match broilers metabolic demands (Wideman et al., 1995). Nitric oxide (NO) is an important endogenous vasodilator produced by endothelial cells using arginine (McQueston et al., 1993; Palmer et. al., 1998), an essential amino acid in birds (Tamil and Ratner, 1963). It has been reported that arginine (ARG) levels supporting maximal growth rate are not adequate for maximal NO production (Taylor et al., 1992; Dietert and Austic, 1994).
A sustained high PAP is associated with hypoxia, endothelial damage and reactive oxygen species (ROS) production. Ascitic broilers have low levels of vitamin E (VE; Bottje et al., 1995) and ascorbic acid (Enkvetchakul et al., 1993) in lung and liver. Vitamin E supplementation reduced the incidence of ascites when administered as a subcutaneous implant but not when provided as a dietary supplement (Bottje et al., 1995, 1997). Vitamin E reacts with organic peroxides interrupting the chain reaction of lipid peroxidation, but in this process some tocopheroxyl radicals are produced, which can act as pro-oxidative agents and initiate oxidative processes (Schneider, 2005).
Ascorbate has been shown to prevent the pro-oxidant activity of a-tocopherol by reducing the a-tocopheroxyl radical to a-tocopherol, then acting as a co-antioxidant (Carr et al., 2000). Through this mechanism, vitamin C (VC) supplementation has been reported to increase or restore plasma and tissue vitamin E (Liu and Lee, 1998; Keller et al., 2004). Furthermore, ascorbate has been reported to have beneficial effects on endothelium -ependent vasodilation by decreasing the levels of superoxide radicals, which inactivate NO (Dudgeon et al., 1998).
The combined effects of ARG, VE and VC on cardiopulmonary performance and ascites parameters have not been studied. We hypothesize that arginine and antioxidant vitamins may have complementary effects on cardiovascular performance, with ARG providing extra substrate for NO production, and vitamins E and C providing protection against oxidative stress. In this study, we evaluated the effects of ARG and either VE or VC, or a combination of ARG, VE and VC, on cardiopulmonary responses after an acute epinephrine challenge and on haematocrit of broiler chickens reared under cold exposure.
Materials and Methods
Ninety-six day-old Cobb 500 male broiler chicks were used in each of two experiments. All broilers were brooded at recommended brooding temperatures until 16 days of age. Thereafter, all broilers were subjected to cool temperature (16°C) until six weeks of age to amplify the incidence of PHS.
All chicks were fed a basal corn-soybean meal diet to meet or exceed the NRC (1994) requirements, including 22.5 per cent CP and 3,150 Kcal/kg of ME (1 to 21 days), or 19 per cent CP and 3,250 Kcal/kg of ME (22 to 42 days). The basal diet included ARG at 1.2 per cent (w/w) and VE at 40IU/kg of feed for both growing periods; the details of the diet composition, along with micronutrients, are described elsewhere (Baurhoo et al., 2007). Birds were divided into four groups: a control group (CTL, basal diet), a high ARG and high VE group (AE, basal diet plus 1per cent supplemental L-arginine monohydrochloride, Sigma; and 200 IU of dl-a-tocopheryl acetate, Rovimix E 50 SD, DSM Nutritional Products), a high ARG and VC group (AC, basal diet plus 1per cent L-arginine; and 500 mg / kg of ascorbic acid, Sigma), and a high ARG, high VE, and VC group (AEC, basal diet plus 1per cent L-arginine, 200 IU of a-tocopherol acetate, and 500mg/kg VC). The diets were not pelleted and were mixed in small batches in order to prevent inactivation of the ascorbic acid.
Surgery
From day 28 to 42, clinically healthy birds were selected for the evaluation of cardio-pulmonary performance (n=7 and n=16 per group for Experiments 1 and 2, respectively). Birds were anaesthetised to a surgical plane with allobarbital. The left brachial artery was isolated and cannulated with 30cm of heparinised polyethylene tubing (PE-50, Becton Dickinson). The left brachial vein was cannulated using 30 cm of heparinised Silastic® tubing (0.012 I.D X 0.025 E.D, VWR International) and the proximal end was advanced through the vein and right ventricle until it reached the pulmonary artery. Both distal ends of the PE-50 polyethylene tubing and the Silastic tubing were attached to blood pressure transducers interfaced with a Trasbridge preamplifier to a Biopac MP100® data acquisition system using Acknowledge® software (Biopac Systems Inc., Goleta, CA 93117) for the continuous measurement of pulmonary arterial pressure (PAP, mm Hg) mean systemic arterial pressure (MAP, mm Hg) and heart rate.
Experimental protocols
Once birds were cannulated, they were allowed to stabilise for 10 minutes. During this period, representative PAP and MAP readings were taken at 300 and 60 seconds before an epinephrine (Epi; 4-(1-Hydroxy-2-[methylamino]ethyl)-1,2-benzenediol hydrochloride; Sigma) challenge (0.5 mg/kg BW, i.v.), to obtain basal values. Epinephrine exerts a strong vasonstrictive effect (Smith et al., 2000) increasing MAP and PAP. Evaluation of the vasodilation capacity was estimated by measuring the increment of the PAP after each challenge and the time that birds within each dietary treatment took to return to basal levels. The PAP response was measured at 30, 60, 120, 300, 600, 720 and 1200 seconds after Epi challenge. All the animal procedures were approved by the institutional animal care committee.
Results and Discussion
The pulmonary arterial relaxation is improved when ARG is supplemented above NRC requirements, and this has been attributed to an increase in NO production (Wideman et al., 1996; Lorenzoni and Ruiz-Feria, 2006). Also, antioxidants such as VE (Bottje et al., 1995) and VC (Xiang et al., 2002), have been shown to reduce PHS mortality. However, the effects of ARG, VE or VC on cardiopulmonary function and ascites incidence are not consistent when used alone (Ruiz-Feria et al., 2001; Lorenzoni and Ruiz-Feria, 2006; Walton et al, 2001). The authors hypothesise that ARG and antioxidant vitamins may have complementary effects on cardiopulmonary performance, with ARG providing extra substrate for NO production, and antioxidant vitamins providing protection against oxidative stress and increasing NO bioavailability.
The basal PAP (before challenge) was not different between clinically healthy broilers in the different treatment groups (Figure 1, -300 and -60 time). There was a significant increase in PAP 30 seconds after EPI challenge within all treatment groups (P<0.05; time 30 versus basal PAP). The PAP after the Epi challenge remained higher compared to the basal levels for up to 300 seconds in the CTL and AE group but less than 300 seconds in the AC and AEC group. Furthermore, when comparing the PAP among treatments at the 30 seconds time (the peak response in PAP), the PAP was lowest (P<0.05) in the AEC group and highest in the CTL group, whereas the AE and AC groups had an intermediate (lower than the CTL group, but higher than the AEC group) and comparable PAP. At the 60, 120 and 300 seconds time, the PAP of the CTL group remained higher than the PAP of the AEC group but not different from the PAP of the AE and AC groups. At 60 seconds after the Epi challenge, the PAP of the AEC group was lower than the PAP of the AC group but not different from the PAP of the AE group. At 120 seconds after the challenge, the PAP of the AEC group was lower when compared with the PAP of both the AE and AC group, but at 300 seconds after the Epi challenge there were no differences among the PAP of the AE, AC, and AEC groups. After the 600 seconds recording the PAP was not different among treatments.
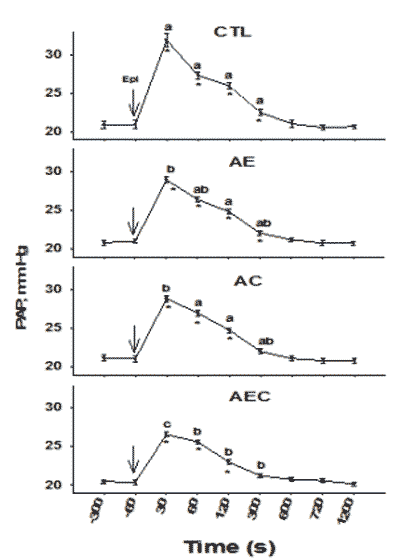
Data were averaged over the representative sample intervals 300 seconds and 60 seconds prior to the epinephrine challenge (basal values) and 30, 60, 120, 300, 600, 720 and 1200 s after Epi injection. Each point represents the mean ± SE. Asterisks represent the values that are different (P<0.05) from all the respective basal values within the same treatment. Letters (a,b) represent values that are different (P<0.05) among treatments at the same sampling time.
Therefore, the birds in the AEC group showed the best pulmonary vasodilation response after an acute EPI challenge, as evidenced by the lower peak in PAP at 30 seconds, the lower PAP from 60 to 300 seconds, and the rapid return of the PAP to basal levels, whereas the birds in the AE and AC group had a better pulmonary vasodilation response than the CTL birds. These results suggest an additive effect of ARG, VE, and VC on cardiopulmonary response. The extra plasma levels of ARG may become available for enzymatic formation of NO, whereas antioxidant vitamins may protect the bioavailability of NO and protect the endothelial integrity, and in this way improving the vasodilator capacity of the lungs.
In that regard, the results also indicate that VE or VC, in combination with ARG, provide limited protection against oxidative stress, but when both antioxidant vitamins are combined, the protective effect is enhanced. The additive effects of VE and VC on antioxidant capacity have been reported in laying hens reared at high temperatures (Sahin et al., 2002), and in apolipoprotein-E-deficient mice (Nespereira et al., 2003; Rodriguez-Gomez et al., 2005). It has been documented that VC ensures a-tocopherol regeneration from a-tocopheroxy radical, thereby preventing the VE pro-oxidant activity, and acting as a 'co-antioxidant' and inhibiting oxidation (Carr et al., 2000). Chickens can synthesise VC but under intensive farming conditions and stressors such as rapid growth, heat or cold, birds may be unable to synthesise adequate amounts of VC (Pardue and Thaxton, 1986). Also, ARG may become a pro-oxidative agent under some circumstances; for instance, higher levels of NO production have been associated with the production of superoxide radicals (Hishikawa and Lüscher, 1997); Ruiz-Feria et al. (2004) reported that high levels of ARG were associated with increased endothelial lesions in the aorta of birds, presumably due to an increased oxidative stress.
The results of these experiments strongly suggest that the combination of VE and VC have additive effects on improving cardiopulmonary performance and reducing pulmonary hypertension, and these may be mediated by reductions in oxidative stress and increased availability of NO.
A higher haematocrit is associated with sustained hypoxia (Yersin et al., 1992) and has been shown to be correlated with ascites susceptibility (Wideman et al., 1998). At three weeks of age (five days after cold exposure started), the haematocrit was higher in the CTL group than in the AEC group but at week 4, there were not differences between groups (Figure 2). However, at weeks 5 and 6, haematocrit values were lowest (P<0.05) in the AEC birds, and highest in the CTL group. Haematocrit values for the AE and AC group were similar between them, lower when compared with the CTL birds but higher when compared with the AEC birds (Figure 2). These haematocrit values were higher than those previously reported by Maxwell et al. (1992) but similar to the ones reported previously by these authors (Lorenzoni and Ruiz-Feria, 2006). The haematocrit values in these experiments match the results of cardiopulmonary performance, further supporting the role of ARG and antioxidants on gas exchange rate and overall cardiopulmonary function in the AEC birds, followed by birds fed ARG and either VE or VC. The mortality attributed to ascites was numerically higher in the CTL group (9/48), followed by the AC group (8/48), the AE group (6/48), and the AEC group (4/48).
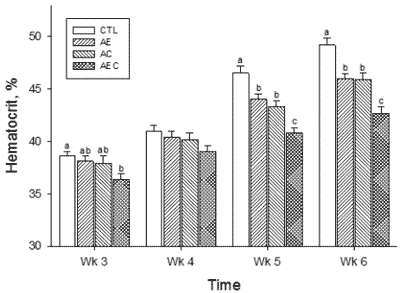
Each bar represents the mean ± SE of 26-27 observations. Bars with different letters within the same age group are statistically different (P<0.05)
Summary
In summary, the concurrent supplementation of ARG, VE and VC significantly improved the pulmonary vasodilation of broiler chickens grown under cold environments after an acute Epi challenge; the improved pulmonary vasodilation was associated with lower haematocrit levels.
Thus, ARG, VE, and VC may have complementary effects on cardiopulmonary performance, by increasing NO bioavailability through a higher substrate for NOS, and presumably by reducing the losses of NO associated with oxidative stress and reducing endothelial damage by NO-derived radicals. Further research is warranted to elucidate these mechanisms.
References
Baurhoo, B., L. Phillip and C.A. Ruiz-Feria. 2007. Effects of purified lignin and mannanoligosaccharides on intestinal integrity and microbial populations in the ceca and litter of broiler chickens. Poult. Sci. 86:1070-1078. Bottje, W., B. Enkvetchakul and R. Moore. 1995. Effect of a-tocopherol on antioxidants, Lipid Peroxidation, and the Incidence of Pulmonary Hypertension Syndrome (Ascites) in Broilers. Poult. Sci. 74: 1356-1369. Bottje, W.G. and R.F. Wideman. 1995. Potential role of free radicals in the pathogenesis of pulmonary hypertension syndrome. Poult. Avian Biol. Rev. 6:221-231. Bottje, W.G., G.F. Erf, T. K.Bersi, S. Wang, D. Barnes and K. W. Beers. 1997. Effect of dietary dl-a-Tocopherol on tissue a-and gamma tocopherol and pulmonary hypertension syndrome (Ascites) in broilers. Poult. Sci. 76:1506-1512. Buys, N., C.W. Scheele, C. Kwakernaak, J.D. Van Der Klis and E. Decuypere. 1999. Performance and physiological variables in broiler chicken lines differing in susceptibility to the ascites syndrome: 1. Changes in blood gases as a function of ambient temperature. Brit. Poult. Sci. 40: 135-139. Carr, A.C., B.Z. Zhu and B. Frei. 2000. Potential antiatherogenic mechanisms of ascorbate (vitamin C) and a-tocopherol (vitamin E). Circ. Res. 87:349-354. Dietert, R.R. and R.E. Austic. 1994. Environment–immune interactions. Poult. Sci. 73: 1062-1076. Dudgeon, S., D.P. Benson, A. MacKenzie, K. Paisley-Zyskiewicz and W. Martin. 1998. Recovery by ascorbate of impaired nitric oxide-dependent relaxation resulting from oxidant stress in rat aorta. Br. J. Pharmacol. 125:782-786. Enkvetchakul, B., W. Bottje, N. Anthony, R. Moore and W. Huff. 1993. Compromised antioxidant status associated with ascites in broilers. Poult. Sci. 72:2272-2280. Hishikawa K, and T.F. Lüscher. 1997. Pulsatile stretch stimulates superoxide production in human aortic endothelial cells. Circulation. 96:3610-6. Keller, U., C. Brandsch and K. Eder. 2004. Supplementation of vitamins C and E increases the vitamin E status but does not prevent the formation of oxysterols in the liver of guinea pigs fed an oxidised fat. Eur. J. Nutr. 43:353–359. Liu, J.F. and Y.W. Lee. 1998. Vitamin C supplementation restores the impaired vitamin E status of guinea pigs fed oxidized frying oil. J. Nutr. 128: 116–122. Lorenzoni, G. and C.A. Ruiz-Feria. 2006. Effects of Vitamin E and L-arginine on Cardiopulmonary function and ascites parameters in broilers chickens reared under sub-normal temperatures. Poult. Sci. 85:2241-2250. Maxwell, M.H., G.W. Robertson and C.C. McCorquodale. 1992. Whole blood and plasma viscosity values in normal and ascitic broiler chickens. Br. Poult. Sci. 33, 871-877. Maxwell M.H. and G.W. Robertson. 1998. UK survey of broiler ascites and sudden death syndromes in 1993. Brit. Poult. Sci. 39:203-215. McQueston, J.A., D.N. Cornfield, I.F. McMurtry and S.H. Abman. 1993. Effects of oxygen and exogenous L-arginine on EDRF activity in fetal pulmonary circulation. Am. J. Physiol. 264:H865-H871. National Research Council. 1994. Nutrient Requirements of Poultry. 9th rev. ed. National Academy Press, Washington, DC. Nespereira, B., M. Perez-Ilzarbe, P. Fernandez, A.M. Fuentes, J.A. Paramo and J.A. Rodriguez. 2003. Vitamin C and E downregulates vascular VEFG and VEGFR-2 expression in apolipoprotein-E-defficient mice. Atherosclerosis. 171:67-73. Palmer, L.A., G.L. Semenza, M.H. Stoler and R.A. Johns. 1998. Hypoxia induces type II NOS gene expression in pulmonary artery endothelial cells via HIF-1. Am. J. Physiol. Lung Cell Mol. Physiol. 274: 212-219. Pardue, S.L. and J.P. Thaxton. 1986. Ascorbic acid in poultry: a review. World's Poult. Sci. J. 42: 107–123. Peacock, A.J., C. Pickett, K. Morris and J.T. Reeves. 1990. Spontaneous hypoxemia and right ventricular hypertrophy in fast growing broiler chickens reared at sea level. Comp. Biochem. Physiol. A. 97:537-541. Ruiz-Feria, C.A., M.T. Kidd and R.F. Wideman. 2001. Plasma levels of arginine, ornithine, and growth performance of broilers fed supplemented L-arginine during cool temperature exposure. Poult. Sci. 80:358-369. Ruiz-Feria, C.A., Y. Yang and H. Nishimura. 2004. Do incremental increases in blood pressure elicit neointimal plaques through endothelial injury? Am. J. Physiol. 287:R1486-R1493. Rodriguez-Gomez, I., R. Wangensteen, J.M. Moreno, V. Chamorro, A. Osuna and F. Vargas. 2005. Effects of chronic inhibition of inducible nitric oxide synthase in hyperthyroid rats. Am. J. Physiol. Endocrinol. Metab. 288:E1252-E1257. Sahin, K., N. Sahin and S. Yaralioglu. 2002. Effects of vitamin C and vitamin E on lipid peroxidation, blood serum metabolitesand mineral concentrations of laying hens reared at high ambient temperature. Biol. Trace Elem. Res. 85:35-45. Scheele, C.W., W. De Wit, M.T. Frankenhuis and P.F. Vereijken. 1991. Ascites in broilers. 1. Experimental factors evoking symptoms related to Ascites. Poult. Sci. 70: 1069-1083. Schneider, C. 2005. Chemistry and biology of vitamin E. Mol. Nutr. Food Res. 49:7-30. Tamil, H. and S. Ratner. 1963. A study of ornithine, citrulline and arginine synthesis in growing chicks. Arch. Biochem. Biophys. 102:259-269. Taylor, R.L., R.E. Austic and R. Dietert. 1992. Dietary arginine influences Rous sarcoma Growth in a major histocompatibility B complex progressor genotype. Proc. Soc. Exp. Biol. Med. 199:38-41. Walton, J.P., R.J. Julian and E.J. Squires. 2001. The effects of dietary flax oil and antioxidants on ascites and pulmonary hypertension in broilers using a low temperature model. Brit. Poult. Sci., 42:123-129. Wideman, R.F. 1988. Ascites in Poultry. Mansato Nutr. Update 6(2):1-7. Wideman, R.F. Jr. 1999. Cardiac output in four-, five- and six-week-old broilers, and hemodynamic responses to intravenous injections of epinephrine. Poult. Sci. 78:392-403. Wideman, R.F., Y.K. Kirby, M. Ismalil, W. Bottje, R. Moore and R. Vardeman. 1995. Supplemental L-arginine Attenuates Pulmonary Hypertension Syndrome (Ascites) in Broilers. Poult. Sci. 74:323-330. Widemann, R.F., Y.K. Kirby, C.D. Tackett, N.E. Marson and R.W. Mcnew. 1996. Cardio-pulmonary function during acute unilateral occlusion of the pulmonary artery in broilers fed diets containing normal or high levels of arginine-HCL. Poult. Sci. 75:1587-1602. Wideman, R.F. Jr, T. Wing, Y.K. Kirby, M.F. Forman, N. Marson, C.D. Tackett and C.A. Ruiz-Feria. 1998. Evaluation of minimally invasive indices for predicting ascites susceptibility in three successive hatches of broilers exposed to cool temperatures. Poult. Sci. 77:1565-1573. Wideman, R.F. and C.D. Tackett. 2000. Cardio-Pulmonary Function in Broilers Reared at warm or Cool Temperatures: Effect of Acute Inhalation of 100 per cent oxygen. Poultry Sci. 79: 257-264. Xiang R.P., W.D. Sun, J.Y. Wang and X.L. Wang. 2002. Effect of vitamin C on pulmonary hypertension and muscularisation of pulmonary arterioles in broilers. Brit. Poult. Sci. 43: 705-712. Yersin, A.G., W.E. Huff, L.F. Kubena, M.H. Elissalde, R.B. Harvey, D.A. Witzel and L.E. Giroir. 1992. Changes in hematological, blood gas, and serum biochemical variables in broilers during exposure to simulated high altitude. Avian Dis. 36:189-196. This paper was first published on FeedInfo.August 2010








